Cardiovascular – Heart
- Heart Intro
- Guidelines
- Exam Protocols
- Image Gallery
- Bibliography
Contrast-enhanced ultrasound (CEUS) can dramatically improve the detection of coronary artery disease and stratify risk of heart attack or stroke.
CEUS is safe, accurate, completely avoids exposure to ionizing radiation, and can change patient outcomes and save lives. In addition, by salvaging non-diagnostic ultrasound examination, CEUS can help avoid unnecessary downstream tests and lower overall health care costs. Further, like conventional ultrasound examinations, CEUS is portable and easily accessible in a variety of clinical settings, provides real time diagnostic information that can speed access to appropriate therapy, and may improve workflows and efficiencies.
CEUS uses ultrasound contrast agents (UCAs), also known as ultrasound enhancement agents, to improve the clarity and reliability of conventional ultrasound scans. UCAs are comprised of liquid suspensions of biodegradable gas-filled microspheres (sometimes called “microbubbles”). When they are injected into a patient’s arm vein during an ultrasound exam, they flow through the body’s microcirculation without impediment, and are metabolized and expelled from the body within minutes.
Unlike agents used for other types of medical imaging, UCAs do not contain dye or present a risk of kidney damage.
UCAs are now integral components of ultrasound imaging throughout the world, and are approved for use in adult and pediatric imaging by the US Food and Drug Administration, the European Medicines Agency, and their counterparts in Asia, the Americas and elsewhere.
FDA-approved cardiac applications are limited to evaluation of the left ventricle of the heart and endocardial borders. However, CEUS scans may have significant diagnostic value with no added safety risk when used off-label during cardiac stress echocardiography and perfusion imaging studies. The Intersocietal Accreditation Commission guidelines for accrediting adult echocardiography laboratories requires the use of an UCA during stress echocardiography under certain specified conditions.
Under certain circumstances, CEUS may be superior to nuclear imaging or conventional ultrasound without a contrast agent. Nuclear scans generally have poorer spatial and temporal resolution and expose patients to ionizing radiation.
ESC(European Society of Cardiology) Guideline 2024 on Chronic Coronary Syndrome
European Association of Cardiovascular Imaging (EACVI) Contrast Echo Recommendations (2017)
Accreditation standards for adult echocardiography laboratories in the U.S.
CEUS cardiac exam protocols International Contrast Ultrasound Society (ICUS) recommendations
Guidelines for the Cardiac Sonographer in the Performance of Contrast Echocardiography
Contrast Echocardiography: The Bubbles and How We Image Them (ASE)
The Basics of Bubbles: What the Clinician Needs to Know (ASE):
The following objectives and protocols for 2022 (Portuguese edition available here and Mandarin edition available here) reflect clinical CEUS practice in many medical centers throughout the world.
Heart Endocardial Border Delineation
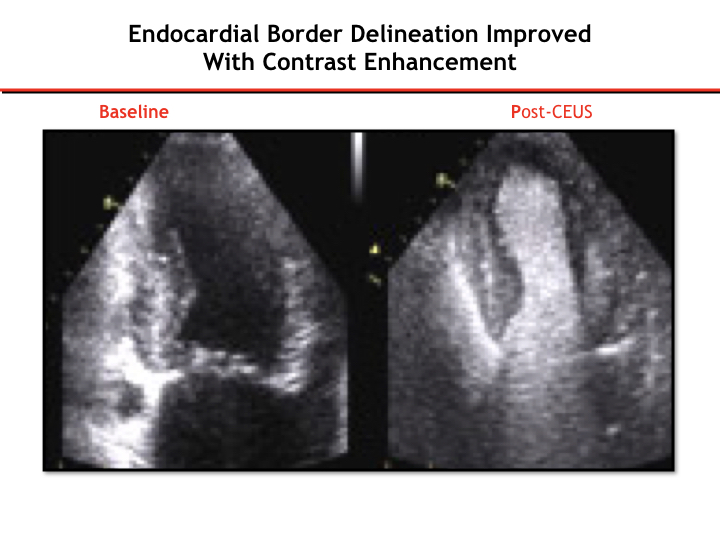
- Mulvagh SL, Rakowski H, Vannan MA, Abdelmoneim SS, Becher H, Bierig SM, et al. ASE consensus statement on the clinical applications of ultrasonic contrast agents in echocardiography. J Am Soc Echocardiogr 2008;21:1179-201.
- GE Healthcare. Optison. Available at: https://promo.gelifesciences.com/gl/ OPTISONIMAGING/misc/Updated-Optison-PI-10-21-14.pdf. Accessed December 2, 2017.
- Lantheus Medical Imaging. Definity. Available at: http://www. definityimaging.com/pdf/DEFINITY_US_PI_515987-0117.pdf. Accessed December 2, 2017.
- Bracco Diagnostics. Lumason. Available at: http://imaging.bracco.com/ sites/braccoimaging.com/files/technica_sheet_pdf/us-en-2017-01-04- spc-lumason.pdf. Accessed December 2, 2017.
- Appis AW, Tracy MJ, Feinstein SB. Update on the safety and efficacy of commercial ultrasound contrast agents in cardiac applications. Echo Res Pract 2015;2:R55-62.
- Intersocietal Accreditation Commission. IAC standards and guidelines for adult echocardiography accreditation. Available at: https://www.intersocietal.org/echo/standards/IACAdultEchocardiographyStandards2017.pdf. Accessed December 3, 2017.
- Aggeli C, Giannopoulos G, Roussakis G, Christoforatou E, Marinos G, Toli C, et al. Safety of myocardial flash contrast echocardiography in combination with dobutamine stress testing for the detection of ischaemia in 5250 studies. Heart 2008;94:1571-7.
- GabrielRS, SmythYM, MenonV, KleinAL, GrimmRS, ThomasJD, etal. Safety of ultrasound contrast agents in stress echocardiography. Am J Cardiol 2008;102:1269-72.
- HerzogCA.Incidence of adverse events associated with use of perflutren contrast agents for echocardiography. JAMA 2008;299:2023-5.
- Kusnetzky LL, Khalid A, Khumri TM, Moe TG, Jones PG, Main ML. Acute mortality in hospitalized patients undergoing echocardiography with and without an ultrasound contrast agent: results in 18,671 consecutive studies. J Am Coll Cardiol 2008;51:1704-6.
- Main ML, Ryan AC, Davis TE, Albano MP, Kusnetzky LL, Hibberd M. Acute mortality in hospitalized patients undergoing echocardiography with and without an ultrasound contrast agent (multicenter registry re- sults in 4,300,966 consecutive patients). Am J Cardiol 2008;102:1742-6.
- Shaikh K, Chang SM, Peterson L, Rosendahl-Garcia K, Quinones MA, Nagueh SF, et al. Safety of contrast administration for endocardial enhancement during stress echocardiography. Am J Cardiol 2008;102: 1444-50.
- Wei K, Mulvagh SL, Carson L, Davidoff R, Gabriel R, Grimm R, et al. The safety of Definity and Optison for ultrasound image enhancement: a retrospective analysis of 78,383 administered contrast doses. J Am Soc Echocardiogr 2008;21:1202-6.
- Abdelmoneim SS, Bernier M, Scott CG, Dhoble A, Ness SA, Hagen ME, et al. Safety of contrast agent use during stress echocardiography: a 4 year experience from a single-center cohort study of 26,774 patients. JACC Cardiovasc Imaging 2009;2:1048-56.
- AnantharamB,ChahalN,ChelliahR,RamzyI,GaniF,SeniorR.Safetyof contrast in stress echocardiography in stable patients and in patients with suspected acute coronary syndrome but negative 12-hour troponin. Am J Cardiol 2009;104:14-8.
- Dolan MS, Gala SS, Dodla S, Abdelmoneim SS, Xie F, Cloutier D, et al. Safety and efficacy of commercially available ultrasound contrast agents for rest and stress echocardiography: a multicenter experience. J Am Coll Cardiol 2009;53:32-8.
- Abdelmoneim SS, Bernier M, Scott CG, Dhoble A, Ness SA, Hagen ME, et al. Safety of contrast use during stress echocardiography in patients with elevated right ventricular systolic pressure: a cohort study. Circ Car- diovasc Imaging 2010;3:240-8.
- ExuzidesA,MainML,ColbyC,GrayburnPA,FeinsteinSB,GoldmanJH. A retrospective comparison of mortality in critically ill hospitalized pa- tients undergoing echocardiography with and without an ultrasound contrast agent. JACC Cardiovasc Imaging 2010;3:578-85.
- Goldberg YH, Ginelli P, Siegel R, Ostfeld RJ, Schaefer M, Spevack DM. Administration of perflutren contrast agents during transthoracic echo- cardiography is not associated with a significant increase in acute mortal- ity risk. Cardiology 2012;122:119-25.
- Weiss RJ, Ahmad M, Villanueva F, Schmitz S, Bhat G, Hibberd M. CaRES (Contrast Echocardiography Registry for Safety Surveillance): a prospec- tive multicenter study to evaluate the safety of the ultrasound contrast agent Definity in clinical practice. J Am Soc Echocardiogr 2012;25:790-5.
- Wever-Pinzon O, Suma V, Ahuja A, Romero J, Sareen N, Henry SA, et al. Safety of echocardiographic contrast in hospitalized patients with pulmo- nary hypertension: a multi-center study. Eur Heart J Cardiovasc Imaging 2012;13:857-62.
- Platts DG, Luis SA, Roper D, Burstow D, Call T, Forshaw A, et al. The safety profile of perflutren microsphere contrast echocardiography dur- ing rest and stress imaging: results from an Australian multicentre cohort. Heart Lung Circ 2013;22:996-1002.
- Main ML, Hibberd MG, Ryan A, Lowe TJ, Miller P, Bhat G. Acute mor- tality in critically ill patients undergoing echocardiography with or without an ultrasound contrast agent. JACC Cardiovasc Img 2014;7:408
- Wei K, Shah S, Jaber WA, DeMaria A. An observational study of the occurrence of serious adverse reactions among patients who receive Op- tison in routine medical practice. J Am Soc Echocardiogr 2014;27: 1006-10.
- Muskula P, Main M. Safety with echocardiographic contrast agents. Circ Cardiovasc Imaging 2017;10:e005459.
- Kurt M, Shaikh KA, Peterson L, Kurrelmeyer KM, Shah G, Nagueh SF, et al. Impact of contrast echocardiography on evaluation of ventricular function and clinical management in a large prospective cohort. J Am Coll Cardiol 2009;53:802-10.
- Senior R, Moreo A, Gaibazzi N, Agati L, Tiemann K, Shivalkar B, et al. Comparison of sulfur hexafluoride microbubble (SonoVue)-enhanced myocardial echocardiography to gated single photon emission computer- ized tomography for the detection of significant coronary artery disease: a large European multicenter study. J Am Coll Cardiol 2013;62:1353-61.
- Main ML, Grayburn PA, Lang RM, Goldman JH, Gibson CM, Sherwin P, et al. Effect of Optison on pulmonary artery systolic pressure and pulmo- nary vascular resistance. Am J Cardiol 2013;112:1657-61.
- Wei K, Main ML, Lang RM, Klein A, Angeli S, Panetta C, et al. The effect of Definity on systemic and pulmonary hemodynamics in patients. J Am Soc Echocardiogr 2012;25:584-8.
- Kutty S, Xiao Y, Olson J, Xie F, Danford DA, Erickson CC, et al. Safety and efficacy of cardiac ultrasound contrast in children and adolescents for resting and stress echocardiography. J Am Soc Echocardiogr 2016; 29:655-62.
- Fine NM, Adelmoneim SS, Dichack A, Kushwaha SS, Park SJ, Mulvagh SL. Safety and feasibility of contrast echocardiography for LVAD evaluation. JACC Cardiovasc Imaging 2014;7:4.
- Bennet CE, Tweet MS, Michelena HI, Schears GJ, Mulvagh SL. Safety and feasibility of contrast echocardiography for ECMO evaluation. JACC Cardiovasc Imaging 2017;10:603-4.
- Kalra A, Shroff GR, Erlien D, Gilbertson DT, Herzog CA. Perflutren- based echocardiographic contrast in patients with right-to-left intracar- diac shunts. JACC Cardiovasc Imaging 2014;7:206-7.
- Leong-Poi H, Rim SJ, Le DE, Fisher NG, Wei K, Kaul S. Perfusion versus function: the ischemic cascade in demand ischemia: implications of single-vessel versus multivessel stenosis. Circulation 2002;105:987-92.
- Kaufmann BA, Wei KS, Lindner JR. Contrast echocardiography. Curr Probl Cardiol 2007;32:51-96.
- Rafter P, Phillips P, Vannan MA. Imaging technologies and techniques. Cardiol Clin 2004;22:181-97.
- Halperin JL, Levine GN, Al-Khatib SM, Birtcher KK, Bozkurt B, Brindis RG, et al. Further evolution of the ACC/AHA clinical practice guidelines recommendation classification system. J Am Coll Cardiol 2016;67:1572-4.
- Porter TR, Abdelmoneim S, Belchik JT, McCulloch ML, Mulvagh SL, Olson JJ. Guidelines for the cardiac sonographer in the performance of contrast echocardiography: a focused update from the American Society of Echocardiography. J Am Soc Echocardiogr 2014;27:797-810.
- Dawson D, Kaul S, Peters D, Rinkevich D, Schnell G, Belcik JT, et al. Prog- nostic value of dipyridamole stress myocardial contrast echocardiogra- phy: comparison with single photon emission computed tomography. J Am Soc Echocardiogr 2009;22:954-60.
- Hoffmann R, Barletta G, von Bardeleben S, Vanoverschelde JL, Kasprzak J, Greis C, et al. Analysis of left ventricular volumes and func- tion—a multicenter comparison of cineventriculography, cardiac mag- netic resonance imaging, unenhanced and contrast enhanced 2D and 3D echocardiography. J Am Soc Echocardiogr 2014;27:292-301.
- Senior R, Monaghan M, Main ML, Zamorano JL, Tiemann K, Agati L, et al. Detection of coronary artery disease with perfusion stress echocar- diography using a novel ultrasound imaging agent: two phase 3 interna- tional trials in comparison with radionuclide perfusion imaging. Eur J Echocardiogr 2009;10:26-35.
- Xie F, Dodla S, O’Leary E, Porter TR. Detection of subendocardial ischemia in the left anterior descending coronary artery territory with real-time myocardial contrast echocardiography during dobutamine stress echocardiography. JACC Cardiovasc Img 2008;1:271-8.
- Abdelmoneim SS, Dhoble A, Bernier M, Erwin PJ, Korosoglou G, Senior R, et al. Quantitative myocardial contrast echocardiography dur- ing pharmacological stress for diagnosis of coronary artery disease: a systematic review and meta-analysis of diagnostic accuracy studies. Eur J Echocardiogr 2009;10:813-25.
- Hacker M, Hoyer HX, Uebleis C, Uberfuhr P, Forester S, La Fougere C, et al. Quantitative assessment of cardiac allograft vasculopathy by real- time myocardial contrast echocardiography: a comparison with conven- tional echocardiographic analyses and [Tc99m] sestamibi SPECT. Eur J Echocardiogr 2008;9:494-500.
- Porter TR, Smith LM, Wu J, Thomas D, Haas JT, Mathers DH, et al. Pa- tient outcome following 2 different stress imaging approaches. J Am Coll Cardiol 2013;61:2246-455.
- Miszalski-Jamka T, Kuntz-Hehner S, Schmidt H, Peter D, Miszalski- Jamka K, Hammerstingl C, et al. Myocardial contrast echocardiography enhances long-term prognostic value of supine bicycle stress two- dimensional echocardiography. J Am Soc Echocardiogr 2009;22:1220-7.
- Arnold JR, Karamitsos TD, Pegg TJ, Francis JM, Olszewski R, Searle N, et al. Adenosine stress myocardial contrast echocardiography for the detection of coronary artery disease: a comparison with coronary angiog- raphy and cardiac magnetic resonance. JACC Cardiovasc Imaging 2010; 3:934-43.
- Hayat SA, Dwivedi G, Jacobsen A, Lim TK, Kinsey C, Senior R. Effects of left bundle branch block on cardiac structure, function, perfusion, and perfusion reserve: implications for myocardial contrast echocardiogra- phy versus radionuclide perfusion imaging for the detection of coronary artery disease. Circulation 2008;117:1832-41.
- Lipiec P, Wejner-Mik P, Krzeminska-Pakula M, Kusmierek J, Plachcinska A, Szuminski R, et al. Accelerated stress real-time myocardial contrast echocardiography for the detection of coronary artery disease: comparison with 99mTc single photon emission computed tomography. J Am Soc Echocardiogr 2008;21:941-7.
- Gudmundsson P, Shahgaldi K, Winter R, Dencker M, Kitlinski M, Thorsson O, et al. Head to head comparison of two modalities of perfu- sion adenosine stress echocardiography with simultaneous SPECT. Car- diovasc Ultrasound 2009;7:19.
- Abdelmoneim SS, Dhoble A, Bernier M, Moir S, Hagen ME, Ness SA, et al. Absolute myocardial blood flow determination using real-time myocardial contrast echocardiography during adenosine stress: compar- ison with single-photon emission computed tomography. Heart 2009; 95:1662-8.
- Gaibazzi N, Rigo F, Squeri A, Ugo F, Reverberi C. Incremental value of contrast myocardial perfusion to detect intermediate versus severe coro- nary artery stenosis during stress-echocardiography. Cardiovasc Ultra- sound 2010;8:16-23.
- Gaibazzi N, Rigo F, Reverberi C. Detection of coronary artery disease by combined assessment of wall motion, myocardial perfusion and coro- nary flow reserve: a multiparametric contrast stress-echocardiography study. J Am Soc Echocardiogr 2010;23:1242-50.
- Porter TR, Adolphson M, High RR, Smith LM, Olson J, Erdkamp M, et al. Rapid detection of coronary artery stenoses with real-time perfusion echocardiography during regadenoson stress. Circ Cardiovasc Imaging 2011;4:628-35.
- Gaibazzi N, Reverberi C, Lorenzoni V, Molinaro S, Porter TR. Prognostic value of high-dose dipyridamole stress myocardial contrast perfusion echocardiography. Circulation 2012;126:1217-24.
- Galiuto L, Garramone B, Scara A, Rebuzzi AG, Crea F, La Torre G, et al. The extent of microvascular damage during myocardial contrast echocar- diography is superior to other known indexes of post-infarct reperfusion in predicting left ventricular remodeling. J Am Coll Cardiol 2008;51:552-9.
- Lonnebakken MT, Bleie O, Strand E, Staal EM, Nygard OK, Gerdts E. Myocardial contrast echocardiography in assessment of stable coronary artery disease at intermediate dobutamine-induced stress level. Echocar- diography 2009;26:52-60.
- Hoffmann R, von Bardeleben S, Barletta G, Pasques A, Kasprzak J, Greis C, et al. Analysis of regional left ventricular function using 2D and 3D unenhanced and contrast enhanced echocardiography in com- parison to cineventriculography and cardiac magnetic resonance. A multicenter comparison of methods. Am J Cardiol 2014;113:395-401.
- Wei K, Peters D, Belcik T, Kalvaitis S, Womak L, Rinkevich D, et al. Pre- dictive instrument using contrast echocardiography in patients present- ing to the emergency department with chest pain and without ST- segment elevation. J Am Soc Echocardiogr 2010;23:636-42.
- Wyrick JJ, Kalvaitis S, McConnell J, Rinkevich D, Kaul S, Wei K. Cost- efficiency of myocardial contrast echocardiography in patients present- ing to the emergency department with chest pain of suspected cardiac origin and a nondiagnostic electrocardiogram. Am J Cardiol 2008;102: 649-52.
- Chelliah RK, Hickman M, Kinsey C, Burden L, Senior R. Myocardial contrast echocardiography versus single photon emission computed to- mography for assessment of hibernating myocardium in ischemic cardio- myopathy: preliminary qualitative and quantitative results. J Am Soc Echocardiogr 2010;23:840-7.
- Abdelmoneim SS, Mankad SV, Bernier M, Dhoble A, Hagen ME, Ness SA, et al. Microvascular function in takotsubo cardiomyopathy with contrast echocardiography: prospective evaluation and review of literature. J Am Soc Echocardiogr 2009;22:1249-55.
- Moon J, Cho IJ, Shim CY, Ha JW, Jang Y, Chung N, et al. Abnormal myocardial capillary density in apical hypertrophic cardiomyopathy can be assessed by myocardial contrast echocardiography. Circ J 2010; 74:2166-72.
- Abdelmoneim SS, Mulvagh SL, Xie F, O’Leary E, Adolphson M, Omer MA, et al. Regadenoson stress real time myocardial perfusion echocardiography for detection of coronary artery disease: feasibility and accuracy of two different ultrasound contrast agents. J Am Soc Echo- cardiograph 2015;28:1393-400.
- Wu J, Barton D, Xie F, O’Leary E, Steuter J, Pavlides G, et al. Comparison of fractional flow reserve assessment with demand stress myocardial contrast echocardiography in angiographically intermediate coronary stenosis. Cir Cardiovasc Imaging 2016;9:e004129.
- Thomas D, Xie F, Smith LM, O’Leary E, Smith K, Olson J, et al. Prospec- tive randomized comparison of conventional stress echocardiography and real time perfusion stress echocardiography in detecting significant coronary artery disease. J Am Soc Echocardiogr 2012;25:1207-14.
- Vamvakidou A, Karogiannis N, Tzalamouras V, Parsons G, Young G, Gurunathan S, et al. Prognostic usefulness of contemporary stress echo- cardiography in patients with left bundle branch block and impact of contrast use in improving prediction of outcome. Eur Heart J Cardiovasc Imaging 2017;18:415-21.
- Zacharias K, Ahmed A, Shah BN, Gurunathan S, Young G, Acosta D, et al. Relative clinical and economic impact of exercise echocardiography vs. exercise electrocardiography, as first line investigation in patients without known coronary artery disease and new stable angina: a ran- domized prospective study. Eur Heart J Cardiovasc Imaging 2017;18: 195-202.
- Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiogra- phy in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 2015;28:1-39.
- Paakkanen R, He W, Savu A, Paterson I, Pituskin E, Mirhadi E, et al. Pro- posal for contrast-enhanced 2D echocardiography reference values in fe- males [abstract]. J Am Soc Echocardiogr 2016;B68.
- Hoffmann R, von Bardeleben S, ten Cate R, Borges AC, Kasprzak J, Firschke C, et al. Assessment of systolic left ventricular function: a multi-centre comparison of cineventriculography, cardiac magnetic reso- nance imaging, unenhanced and contrast-enhanced echocardiography. Eur Heart J 2005;26:607-16.
- Thavendiranathan P, Grant AD, Negishi T, Plana JC, Puporvic ZB, Marwick TM. Reproducibility of echocardiographic techniques for the sequential assessment of left ventricular ejection fraction and volumes: application to patients undergoing cancer chemotherapy. J Am Coll Car- diol 2013;61:77-84.
- Hoffmann R, von Bardeleben S, Kasprzak JD, Borges AC, ten Cate F, Firschke C, et al. Analysis of regional left ventricular function by cineven- triculography, cardiac magnetic resonance imaging, and unenhanced and contrast-enhanced echocardiography: a multicenter comparison of methods. J Am Coll Cardiol 2006;47:121-8.
- Saric M, Armour AC, Arnaout MS, Chaudhry FA, Grimm RA, Kronzon I, et al. Guidelines for the use of echocardiography in the eval- uation of cardiac source of embolism. J Am Soc Echocardiogr 2016;29: 1-42.
- Nagueh SF, Bierig SM, Budoff MJ, Desai M, Dilsizian V, Eidem B, et al. American Society of Echocardiography recommendations for multi- modality cardiovascular imaging of patients with hypertrophic cardiomy- opathy: endorsed by the American Society of Nuclear Cardiology, Soci- ety for Cardiovascular Magnetic Resonance, and Society of Cardiovascular Computed Tomography. J Am Soc Echocardiogr 2011; 24:473-98.
- Weinsaft JW, Kim J, Medicheria CB, Ma CL, Codella NC, Kukar N, et al. Echocardiographic algorithm for post myocardial infarction LV thrombus: a gatekeeper for thrombus evaluation by delayed enhance- ment CMR. JACC Cardiovasc Imaging 2016;9:505-15.
- Kirkpatrick JN, Wong T, Bednarz JE, Spencer KT, Sugeng L, Ward RP, et al. Differential diagnosis of cardiac masses using contrast echocardio- graphic perfusion imaging. J Am Coll Cardiol 2004;43:1412-9.
- Duke J, Greaves K, Dettrick A. Use of microbubble contrast in the diag- nosis of a left ventricular papillary fibroelastoma. Echo Res Pract 2015;2: K43-5.
- Bhattacharyya S, Khattar R, Senior R. Characterization of intra-cardiac masses by myocardial contrast echocardiography. Int J Cardiol 2013; 163:e11-3.
- Uenishi EK, Caldas MA, Tsutsui JM, Abduch MC, Sbano JC, Kalil Filho R, et al. Evaluation of cardiac masses by real-time perfusion imaging echo- cardiography. Cardiovasc Ultrasound 2015;13:23.
- Rowin EJ, Maron BJ, Hans TTS, Garberich RF, Wang W, Link MS, et al. Hypertrophic cardiomyopathy with left ventricular apical aneurysm. Im- plications for risk stratification and management. J Am Coll Cardiol 2017; 69:761-73.
- Kebed KY, Al Adham RI, Bishu K, Askew JW, Klarich KW, Oh JK, et al. Evaluation of apical pouches in hypertrophic cardiomyopathy using car- diac MRI. Int J Cardiovasc Imaging 2014;30:591-7.
- Bernier M, Abdelmoneim SS, Stuart Moir W, Eifert Rain SS, Chandrasekaran K, Ammash NM, et al. CUTE-CV: a prospective study of enhanced left atrial appendage visualization with microbubble contrast agent use during transesophageal echocardiography guided car- dioversion. Echocardiography 2013;30:1091-7.
- Jung PH, Mueller M, Schuhmann C, Eickhoff M, Schnieder P, Seemueller G, et al. Contrast enhanced transesophageal echocardiogra- phy in patients with atrial fibrillation referred to electrical cardioversion improves atrial thrombus detection and may reduce thromboembolic events. Cardiovasc Ultrasound 2013;11:1.
- Plana JC, Mikati IA, Dokainish H, Lakkis N, Abukhalil J, Davis R, et al. A randomized cross-over study for evaluation of the effect of image optimi- zation with contrast on the diagnostic accuracy of dobutamine echocar- diography in coronary artery disease: the OPTIMIZE trial. JACC Cardiovasc Imaging 2008;1:145-52.
- Shah BN, Balaji G, Alhajiri A, Ramzy IS, Ahmadvazir S, Senior R. Incre- mental diagnostic and prognostic value of contemporary stress echocar- diography in a chest pain unit: mortality and morbidity outcomes from a real-world setting. Circ Cardiovasc Imaging 2013;6:202-9.
- Jayaweera AR, Wei K, Coggins M, Bin JP, Goodman C, Kaul S. Role of capillaries in determining CBF reserve: new insights using myocardial contrast echocardiography. Am J Physiol 1999;277:H2363-72.
- Wei K, Le E, Bin JP, Coggins M, Jayawera AR, Kaul S. Mechanism of reversible (99m) Tc-sestamibi perfusion defects during pharmacologi- cally induced vasodilatation. Am J Physiol Heart Circ Physiol 2001; 280:H1896-904.
- Mattoso AA, Kowatsch I, Tsutsui JM, de la Cruz VY, Ribeiro HB, Sbano JC, et al. Prognostic value of qualitative and quantitative vasodi- lator stress myocardial perfusion echocardiography in patients with known or suspected coronary artery disease. J Am Soc Echocardiogr 2013;26:539-47.
- Gaibazzi N, Porter T, Lorenzoni V, Pontone G, De Santis D, De Rosa A, et al. Effect of coronary revascularization on the prognostic value of stress myocardial contrast wall motion and perfusion imaging. J Am Heart As- soc 2017;6:e006202.
- Balcells E, Powers ER, Lepper W, Belcik T, Wei K, Ragosta M, et al. Detec- tion of myocardial viability by contrast echocardiography in acute infarc- tion predicts recovery of resting function and contractile reserve. J Am Coll Cardiol 2003;41:827-33.
- Janardhanan R, Moon JC, Pennell DJ, Senior R. Myocardial contrast echocardiography accurately reflects transmurality of myocardial necro- sis and predicts contractile reserve after acute myocardial infarction. Am Heart J 2005;149:355-62.
- Lindner JR, Skyba DM, Goodman NC, Jayaweera AR, Kaul S. Changes in myocardial blood volume with graded coronary stenosis. Am J Physiol 1997;272:H567-75.
- Wei K, Jayaweera AR, Firoozan S, Linka A, Skyba DM, Kaul S. Quantifi- cation of myocardial blood flow with ultrasound-induced destruction of microbubbles administered as a constant venous infusion. Circulation 1998;97:473-83.
- Porter TR, Xie F, Kricsfeld D, Armbruster RW. Improved myocardial contrast with second harmonic transient ultrasound response imaging in humans using intravenous perfluorocarbon-exposed sonicated dextrose albumin. J Am Coll Cardiol 1996;27:1497-501.
- Leong-Poi H, Le E, Rim SJ, Sakuma T, Kaul S, Wei K. Quantification of myocardial perfusion and determination of coronary stenosis severity during hyperemia using real-time myocardial contrast echocardiography. J Am Soc Echocardiogr 2001;14:1173-82.
- Leong-Poi H, Swales J, Jayaweera AR, Bin JP, Kaul S, Lindner JR. Effect of microbubble exposure to ultrasound on quantitation of myocardial perfusion. Echocardiography 2005;22:503-9.
- Wei K, Ragosta M, Thorpe J, Coggins M, Moos S, Kaul S. Noninvasive quantification of coronary blood flow reserve in humans using myocar- dial contrast echocardiography. Circulation 2001;103:2560-5.
- Vogel R, Indermuhle A, Reinhardt J, Meier P, Siegrist PT, Namdar M, et al. The quantification of absolute myocardial perfusion in humans by contrast echocardiography: algorithm and validation. J Am Coll Cardiol 2005;45:754-62.
- Peltier M, Vancraeynest D, Pasquet A, Ay T, Roelants V, D’Hondt AM, et al. Assessment of the physiologic significance of coronary disease with dipyridamole real-time myocardial contrast echocardiography. Comparison with technetium-99m sestamibi single-photon emission computed tomography and quantitative coronary angiography. J Am Coll Cardiol 2004;43:257-64.
- Janardhanan R, Senior R. Accuracy of dipyridamole myocardial contrast echocardiography for the detection of residual stenosis of the infarct- related artery and multivessel disease early after acute myocardial infarc- tion. J Am Coll Cardiol 2004;43:2247-52.
- Senior R, Lepper W, Pasquet A, Chung G, Hoffman R, Vanoverschelde JL, et al. Myocardial perfusion assessment in patients with medium probability of coronary artery disease and no prior myocar- dial infarction: Comparison of myocardial contrast echocardiography with 99mTc single-photon emission computed tomography. Am Heart J 2004;147:1100-5.
- Senior R, Janardhanan R, Jeetley P, Burden L. Myocardial contrast echo- cardiography for distinguishing ischemic from nonischemic first-onset acute heart failure: insights into the mechanism of acute heart failure. Cir- culation 2005;112:1587-93.
- Anantharam B, Janardhanan R, Hayat S, Hickman M, Chahal N, Bassett P, et al. Coronary flow reserve assessed by myocardial contrast echocardiography predicts mortality in patients with heart failure. Eur J Echocardiogr 2011;12:69-75.
- Di Bello V, Giorgi D, Pedrinelli R, Talini E, Palagi C, Delle Donne MG, et al. Early impairment of myocardial blood flow reserve in men with essential hypertension: a quantitative myocardial contrast echocardiogra- phy study. J Am Soc Echocardiogr 2004;17:1037-43.
- Rinkevich D, Belcik T, Gupta NC, Cannard E, Alkayed NJ, Kaul S. Cor- onary autoregulation is abnormal in syndrome X: insights using myocardial contrast echocardiography. J Am Soc Echocardiogr 2013; 26:290-6.
- Schinkel AF, Kaspar M, Staub D. Contrast-enhanced ultrasound: clinical applications in patients with atherosclerosis. Int J Cardiovasc Imaging 2016;32:35-48.
- Johri AM, Herr JE, Li TY, Yau O, Nambi V. Novel ultrasound methods to investigate carotid artery plaque vulnerability. J Am Soc Echocardiogr 2017;30:139-48.
- Ferrer JM, Samso JJ, Serrando JR, Valenzuela VF, Montoya SB, Docampo MM. Use of ultrasound contrast in the diagnosis of carotid artery occlusion. J Vasc Surg 2000;31:736-41.
- Droste DW, Jurgens R, Nabavi DG, Schuierer G, Weber S, Ringelstein EB. Echocontrast-enhanced ultrasound of extracranial internal carotid artery high-grade stenosis and occlusion. Stroke 1999;30:2302-6.
- Staub D, Patel MB, Tibrewala A, Ludden D, Johnson M, Espinosa P, et al. Vasa vasorum and plaque neovascularization on contrast-enhanced ca- rotid ultrasound imaging correlates with cardiovascular disease and past cardiovascular events. Stroke 2010;41:41-7.
- Huang R, Abdelmoneim SS, Ball CA, Nhola LF, Farrell AM, Feinstein S, et al. Detection of carotid atherosclerotic plaque neovascularization using contrast enhanced ultrasound: a systematic review and meta-analysis of diagnostic accuracy studies. J Am Soc Echocardiogr 2016;29:491-502.
- Li C, He W, Guo D, Chen L, Jin X, Wang W, et al. Quantification of ca- rotid plaque neovascularization using contrast-enhanced ultrasound with histopathologic validation. Ultrasound Med Biol 2014;40:1827-33.
- Grewe PH, Mugge A, Germing A, Harrer E, Baberg H, Hanefeld C, et al. Occlusion of pseudoaneurysms using human or bovine thrombin using contrast-enhanced ultrasound guidance. Am J Cardiol 2004;93:1540-2.
- Lindner JR, Womack L, Barrett EJ, Weltman J, Price W, Harthun NL, et al. Limb stress-rest perfusion imaging with contrast ultrasound for the assess- ment of peripheral arterial disease severity. JACC Cardiovasc Imaging 2008;1:343-50.
- Elkouri S, Panneton JM, Andrews JC, Lewis BD, McKusick MA, Noel AA, et al. Computed tomography and ultrasound in follow-up of patients af- ter endovascular repair of abdominal aortic aneurysm. Ann Vasc Surg 2004;18:271-9.
- Bredahl KK, Taudorf M, Lonn L, Vogt KC, Sillesen H, Eiberg JP. Contrast enhanced ultrasound can replace computed tomography angiography for surveillance after endovascular aortic aneurysm repair. Eur J Vasc Endo- vasc Surg 2016;52:729-34.
- Chung J, Kordzadeh A, Prionidis I, Panayiotopoulos Y, Browne T. Contrast-enhanced ultrasound (CEUS) versus computed tomography angiography (CTA) in detection of endoleaks in post-EVAR patients. Are delayed type II endoleaks being missed? A systematic review and meta-analysis. J Ultrasound 2015;18:91-9.
- Wei K. Utility of contrast echocardiography in the emergency depart- ment. JACC Caradiovasc Imaging 2010;3:197-203.
- Douglas PS, Garcia MJ, Haines DE, Lai WW, Manning WJ, Patel AR, et al. ACCF/ASE/AHA/ASNC/HFSA/HRS/SCAI/SCCM/SCMR 2011 appropriate use criteria for echocardiography. A report of the American College of Cardiology Foundation appropriate use criteria task force, American Society of Echocardiography, American Heart Association, American Society of Nuclear Cardiology, Heart Failure Society of Amer- ica, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, Society of Critical Care Medicine, Society of Cardiovascu- lar Computed Tomography, Society for Cardiovascular Magnetic Reso- nance, American College of Chest Physicians. Imaging J Am Soc Echocardiogr 2011;24:229-67.
- Dwivedi G, Janardhanan R, Hayat SA, Swinburn JM, Senior R. Prognostic value of myocardial viability detected by myocardial contrast echocardi- ography early after acute myocardial infarction. J Am Coll Cardiol 2007; 50:327-34.
- Kutty S, Olson J, Danford CJ, Sandene EK, Xie F, Fletcher SE, et al. Ultra- sound contrast and real-time perfusion in conjunction with supine bicy- cle stress echocardiography for comprehensive evaluation of surgically corrected congenital heart disease. Eur Heart J Cardiovasc Imaging 2012;13:500-9.
- McMahon CJ, Ayres NA, Bezold LI, Lewin MB, Alonzo M, Altman CA, et al. Safety and efficacy of intravenous contrast imaging in pediatric echocardiography. Pediatr Cardiol 2005;26:413-7.
- Parker JM, Weller MW, Feinstein LM, Adams RJ, Main M, Grayburn PA, et al. Safety of ultrasound contrast agents in patients with known or sus- pected cardiac shunts. Am J Cardiol 2013;112:1039-45.
- Ryan T, Berlacher K, Lindner JR, Mankad SV, Rose GA, Wang A. CO- CATS 4 Task Force 5: training in echocardiography. J Am Coll Cardiol 2015;65:1786-99.
- Honos G, Amyot R, Choy J, Leong-Poi H, Schnell G, Yu E. Contrast echo- cardiography in Canada: Canadian Cardiovascular Society/Canadian Society of Echocardiography position paper. Can J Cardiol 2007;23: 351-6.
- Tang A, Chiew SK, Rashkovetsky R, Becher H, Choy JB. Feasibility of sonographer-administered echocontrast in a large-volume tertiary-care echocardiography laboratory. Can J Cardiol 2013;29:391-5.
- Castello R, Bella JN, Rovner A, Swan J, Smith J, Shaw L. Efficacy and time- efficiency of a ‘‘sonographer-driven’’ contrast echocardiography protocol in a high-volume echocardiography laboratory. Am Heart J 2003;145: 535-41.
- Xie F, Lof J, Everbach C, He A, Bennett RM, Matsunaga T, et al. Treat- ment of acute intravascular thrombi with diagnostic ultrasound and intra- venous microbubbles. JACC Cardiovasc Imaging 2009;2:511-8.
- Chen X, Leeman JE, Wang J, Pacella JJ, Villanueva FS. New insights into mechanisms of sonothrombolysis using ultra-high-speed imaging. Ultra- sound Med Biol 2014;40:258-62.
- Xie F, Gao S, Wu J, Lof J, Radio S, Vignon F, et al. Diagnostic ultrasound induced inertial cavitation to non-invasively restore coronary and microvascular flow in acute myocardial infarction. PLoS ONE 2013;8: e69780.
- Xie F, Slikkerverr J, Gao S, Lof J, Kamp O, Unger E, et al. Coronary and microvascular thrombolysis with guided diagnostic ultrasound and mi- crobubbles in acute ST segment elevation myocardial infarction. J Am Soc Echocardiogr 2011;24:1400-8.
- Belcik JT, Mott BH, Xie A, Zhao Y, Kim S, Lindner NJ, et al. Augmenta- tion of limb perfusion and reversal of tissue ischemia produced by ultrasound-mediated microbubble cavitation. Circ Cardiovasc Imaging 2015;8:e002979.
- Mathias W Jr., Tsutsui JM, Tavares B, Xie F, Aguiar MO, Garcia DR, et al. Diagnostic ultrasound impulses improve microvascular flow in patients with STEMI receiving intravenous microbubbles. J Am Coll Cardiol 2016;67:2506-15.
- Nahrendorf M, Sosnovik DE, French BA, Swirski FK, Bengel F, Sadeghi MM, et al. Multimodality cardiovascular molecular imaging, part II. Circ Cardiovasc Imaging 2009;2:56-70.
- Lindner JR. Molecular imaging of cardiovascular disease with contrast- enhanced ultrasonography. Nat Rev Cardiol 2009;6:475-81.
- Mott B, Packwood W, Xie A, Belcik JT, Taylor RP, Zhao Y, et al. Echocar- diographic ischemic memory imaging through complement-mediated vascular adhesion of phosphatidylserine-containing microbubbles. JACC Cardiovasc Imaging 2016;9:934-46.
- Maul TM, Dudgeon DD, Beste MT, Hammer DA, Lazo JS, Villanueva FS, et al. Optimization of contrast ultrasound agents with computational models to improve selection of ligands and binding strength. Biotechnol Bioeng 2010;107:854-64.
- Leng X, Wang J, Carson A, Chen X, Fu H, Ottoboni S, et al. Ultrasound detection of myocardial ischemic memory using an E-selectin target- ing peptide amenable to human application. Mol Imaging 2014;16: 1-9.
- Kaufmann BA, Carr CL, Belcik JT, Xie A, Yue Q, Chadderdon S, et al. Molecular imaging of the initial inflammatory response in atheroscle- rosis: Implications for early detection of disease. Arterioscler Thromb Vasc Biol 2010;30:54-9.
- Liu Y, Davidson BP, Yue Q, Belcik T, Xie A, Inaba Y, et al. Molecular im- aging of inflammation and platelet adhesion in advanced atherosclerosis effects of antioxidant therapy with nadph oxidase inhibition. Circ Cardi- ovasc Imaging 2013;6:74-82.
- Hamilton AJ, Huang SL, Warnick D, Rabbat M, Kane B, Nagaraj A, et al. Intravascular ultrasound molecular imaging of atheroma components in vivo. J Am Coll Cardiol 2004;43:453-60.
- Winter PM, Caruthers SD, Allen JS, Cai K, Williams TA, Lanza GM, et al. Molecular imaging of angiogenic therapy in peripheral vascular disease with alpha-beta3-integrin-targeted nanoparticles. Magn Reson Med 2010;64:369-76.
- Wu W, Zhang Z, Zhuo L, Zhou L, Liu P, He Y, et al. Ultrasound molec- ular imaging of acute cellular cardiac allograft rejection in rat with t-cell- specific nanobubbles. Transplantation 2013;96:543-9.
- Weller GE, Lu E, Csikari MM, Klibanov AL, Fischer D, Wagner WR, et al. Ultrasound imaging of acute cardiac transplant rejection with microbub- bles targeted to intercellular adhesion molecule-1. Circulation 2003;108: 218-24.
- Steinl DC, Xu L, Khanicheh E, Ellertsdottir E, Ochoa-Espinosa A, Mitterhuber M, et al. Noninvasive contrast-enhanced ultrasound molec- ular imaging detects myocardial inflammatory response in autoimmune myocarditis. Circ Cardiovasc Imaging 2016;9:e004720.
- Villanueva FS, Lu E, Bowry S, Kilic S, Tom E, Wang J, et al. Myocardial ischemic memory imaging with molecular echocardiography. Circulation 2007;115:345-52.
- Davidson BP, Kaufmann BA, Belcik JT, Xie A, Qi Y, Lindner JR. Detec- tion of antecedent myocardial ischemia with multiselectin molecular im- aging. J Am Coll Cardiol 2012;60:1690-7.
- Ryu JC, Davidson BP, Xie A, Qi Y, Zha D, Belcik JT, et al. Molecular im- aging of the paracrine proangiogenic effects of progenitor cell therapy in limb ischemia. Circulation 2013;127:710-9.
- Shim CY, Liu YN, Atkinson T, Xie A, Foster T, Davidson BP, et al. Mo- lecular imaging of platelet-endothelial interactions and endothelial von Willebrand factor in early and mid-stage atherosclerosis. Circ Cardiovasc Imaging 2015;8:e002765.
- Wang J, Qin B, Chen X, Wagner WR, Villanueva FS. Ultrasound molecu- lar imaging of angiogenesis using vascular endothelial growth factor- conjugated microbubbles. Mol Pharm 2017;14:781-90.
- Wang X, Hagemeyer CE, Hohmann JD, Leitner E, Armstrong PC, Jia F, et al. Novel single-chain antibody-targeted microbubbles for molecular ultrasound imaging of thrombosis: validation of a unique noninvasive method for rapid and sensitive detection of thrombi and monitoring of success or failure of thrombolysis in mice. Circulation 2012;125:3117-26.
- Xie F, Lof J, Matsunaga T, Zutshi R, Porter TR. Diagnostic ultrasound combined with glycoprotein IIb/IIIa-targeted microbubbles improves microvascular recovery after acute coronary thrombotic occlusions. Cir- culation 2009;119:1378-85.
- Kaya M, Toma C, Wang J, Grata M, Fu H, Villanueva FS, et al. Acoustic radiation force for vascular cell therapy: in vitro validation. Ultrasound Med Biol 2012;38:1989-97.
- Toma C, Fisher A, Wang J, Chen X, Grata M, Leeman J, et al. Vascular endoluminal delivery of mesenchymal stem cells using acoustic radiation force. Tissue Eng J Part A 2011;17:1457-64.
- Xie A, Belcik T, Qi Y, Morgan TK, Champaneri SA, Taylor S, et al. Ultra- sound-mediated vascular gene transfection by cavitation of endothelial- targeted cationic microbubbles. JACC Cardiovasc Imaging 2012;5: 1253-62.
- Sun L, Huang CW, Wu J, Chen KJ, Li SH, Weisel RD, et al. The use of cationic microbubbles to improve ultrasound-targeted gene delivery to the ischemic myocardium. Biomaterials 2013;34:2107-16.
- Xie A, Wu MD, Cigarroa G, Belcik JT, Ammi A, Moccetti F, et al. Influ- ence of DNA-microbubble coupling on contrast ultrasound-mediated gene transfection in muscle and liver. J Am Soc Echocardiogr 2016;29: 812-8.
- Christiansen JP, French BA, Klibanov AL, Kaul S, Lindner JR. Targeted tissue transfection with ultrasound destruction of plasmid-bearing cationic microbubbles. Ultrasound Med Biol 2003;29:1759-67.
- Juffermans LJ, Dijkmans PA, Musters RJ, Visser CA, Kamp O. Transient permeabilization of cell membranes by ultrasound-exposed microbub- bles is related to formation of hydrogen peroxide. Am J Physiol Heart Circ Physiol 2006;291:H1595-601.
- Helfield B, Chen X, Qin B, Watkins S, Villanueva FS. Biophysical insight into mechanisms of sonoporation. Proc Natl Acad Sci U S A 2016;113: 9983-8.
- Chen S, Shohet RV, Bekeredjian R, Frenkel P, Grayburn PA. Optimiza- tion of ultrasound parameters for cardiac gene delivery of adenoviral or plasmid deoxyribonucleic acid by ultrasound-targeted microbubble destruction. J Am Coll Cardiol 2003;42:301-8.
- Bekeredjian R, Chen S, Frenkel PA, Grayburn PA, Shohet RV. Ultra- sound-targeted microbubble destruction can repeatedly direct highly specific plasmid expression to the heart. Circulation 2003;108:1022-6.
- Leong-Poi H, Kuliszewski MA, Lekas M, Sibbald M, Teichert- Kuliszewska K, Klibanov AL, et al. Therapeutic arteriogenesis by ultrasound-mediated VEGF165 plasmid gene delivery to chronically ischemic skeletal muscle. Circ Res 2007;101:295-303.
- Fujii H, Matkar P, Liao C, Rudenko D, Lee PJ, Kuliszewski MA, et al. Opti- mization of ultrasound-mediated anti-angiogenic cancer gene therapy. Mol Ther Nucleic Acids 2013;2:e94.
- Shohet RV, Chen S, Zhou YT, Wang Z, Meidell RS, Unger RH, et al. Echocardiographic destruction of albumin microbubbles directs gene de- livery to the myocardium. Circulation 2000;101:2554-6.
- Kopechek JA, Carson AR, McTiernan CF, Chen X, Klein EC, Villanueva FS. Cardiac gene expression knockdown using small inhibi- tory RNA-loaded microbubbles and ultrasound. PLoS One 2016;11: e0159751.
- Cao WJ, Rosenblat JD, Roth NC, Kuliszewski MA, Matkar PN, Rudenko D, et al. Therapeutic angiogenesis by ultrasound-mediated mi- croRNA-126-3p delivery. Arterioscler Thromb Vasc Biol 2015;35: 2401-11.
- Fujii H, Li S-H, Wu J, Miyagi Y, Yau TM, Rakowski H, et al. Repeated and targeted transfer of angiogenic plasmids into the infarcted rat heart via ultrasound targeted microbubble destruction enhances cardiac repair. Eur Heart J 2011;32:2075-84.
- Fujii H, Sun Z, Li SH, Wu J, Fazel S, Weisel RD, et al. Ultrasound-targeted gene delivery induces angiogenesis after a myocardial infarction in mice. JACC Cardiovascular Imaging 2009;2:869-79.
- Lee PJ, Rudenko D, Kuliszewski MA, Liao C, Kabir MG, Connelly KA, et al. Survivin gene therapy attenuates left ventricular systolic dysfunction in doxorubicin cardiomyopathy by reducing apoptosis and fibrosis. Car- diovasc Res 2014;101:423-33.
- Chen S, Chen J, Huang P, Meng XL, Clayton S, Shen JS, et al. Myocardial regeneration in Adriamycin cardiomyopathy by nuclear expression of GLP1 using ultrasound targeted microbubble destruction. Biochem Bio- phy Res Commun 2015;458:823-9.
- Chen S, Shimoda M, Chen J, Grayburn PA. Stimulation of adult resident cardiac progenitor cells by durable myocardial expression of thymosin beta 4 with ultrasound-targeted microbubble delivery. Gene Ther 2013;20:225-33.
- Smith AH, Kuliszewski MA, Liao C, Rudenko D, Stewart DJ, Leong- Poi H. Sustained improvement in perfusion and flow reserve after tempo- rally separated delivery of vascular endothelial growth factor and angiopoietin-1 plasmid deoxyribonucleic acid. J Am Coll Cardiol 2012; 59:1320-8.
- Chen S, Bastarrachea RA, Roberts BJ, Voruganti VS, Frost PA, Nava-Gonzalez EJ, et al. Successful beta cells islet regeneration in streptozotocin-induced diabetic baboons using ultrasound-targeted mi- crobubble gene therapy with cyclinD2/CDK4/GLP1. Cell Cycle 2014; 13:1145-51.
- Kuliszewski MA, Kobulnik J, Lindner JR, Stewart DJ, Leong-Poi H. Vascular gene transfer of SDF-1 promotes endothelial progenitor cell engraftment and enhances angiogenesis in ischemic muscle. Mol Ther 2011;19:895-902.
- Chen HH, Matkar PN, Afrasiabi K, Kuliszewski MA, Leong-Poi H. Pros- pect of ultrasound-mediated gene delivery in cardiovascular applications. Expert Opin Biol Ther 2016;16:815-26.
- Unger E, Porter T, Lindner J, Grayburn P. Cardiovascular drug delivery with ultrasound and microbubbles. Adv Drug Deliv Rev 2014;72:110-26.
- Belcik JT, Davidson BP, Xie A, Wu MD, Yadava M, Qi Y, et al. Augmen- tation of muscle blood flow by ultrasound cavitation is mediated by ATPand purinergic signaling. Circulation 2017;135:1240-52.
- Baibhav B, Mahabir CA, Xie F, Shostrom VK, McCashland TM, Porter TR. Predictive value of dobutamine stress perfusion echocardiog- raphy in contemporary end-stage liver disease. J Am Heart Assoc 2017;6: e005102.
- Chamsi-Pasha MA, Xie F, Smith LM, Miles C, Porter TR. Prognostic value of demand stress real time perfusion imaging in patients with advanced kidney disease undergoing renal transplantation. JACC Cardiovasc Img 2017;10:1528-9.
- Weissman IA, Dickinson CZ, Dworkin HJ, O’Neill WW, Juni JE. Cost- effectiveness of myocardial perfusion imaging with SPECT in the emer- gency department evaluation of patients with unexplained chest pain. Radiology 1996;199:353-7.
- Des Prez RD, Shaw LJ, Gillespie RL, Jaber WA, Noble GL, Soman P, Wolinsky DG, et al. Cost-effectiveness of myocardial perfusion imaging: a summary of the currently available literature. J Nucl Cardiol 2005;12: 750-9.
- Tong KL, Kaul S, Wang XQ, Rinkevich D, Kalvaitis S, Belcik T, et al. Myocardial contrast echocardiography versus thrombolysis in myocar- dial infarction score in patients presenting to the emergency department with chest pain and a nondiagnostic electrocardiogram. J Am Coll Car- diol 2005;46:920-7.
- Achenbach S, Andreotti F, Arden C, Budaj A, Bugiardini R, Crea F, et al. 2013 ESC guidelines on the management of stable coronary artery dis- ease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J 2013;34:2949-3003.
- Xiu J, Cui K, Wang Y, Zheng H, Chen G, Feng Q, et al. Prognostic value of myocardial perfusion analysis in patients with coronary artery disease: a meta-analysis. J Am Soc Echocardiogr 2017;30:270-81.
- Harrrison SD, Harrison MA, DuVall WL. Stress myocardial perfusion im- aging in the emergency department-new techniques for speed and diag- nostic accuracy. Curr Cardiol Rev 2012;8:116-22.
- Thomas R. Porter, MD, FASE (Chair), Sharon L. Mulvagh, MD, FASE (Co-Chair), Sahar S. Abdelmoneim, MBBCH, MSc, MS, FASE, Harald Becher, MD, PhD, J. Todd Belcik, BS, ACS, RDCS, FASE, Michelle Bierig, MPH, ACS, RDCS, FASE, Jonathan Choy, MD, MBA, FASE, Nicola Gaibazzi, MD, PhD, Linda D. Gillam, MD, MPH, FASE, Rajesh Janardhanan, MD, MRCP, FASE, Shelby Kutty, MD, PhD, MHCM, FASE, Howard Leong-Poi, MD, FASE, Jonathan R. Lindner, MD, FASE, Michael L. Main, MD, FASE, Wilson Mathias, Jr., MD, Margaret M. Park, BS, ACS, RDCS, RVT, FASE, Roxy Senior, MD, DM, and Flordeliza Villanueva, MD Clinical Applications of Ultrasonic Enhancing Agents in Echocardiography: 2018 American Society of Echocardiography Guidelines Update






